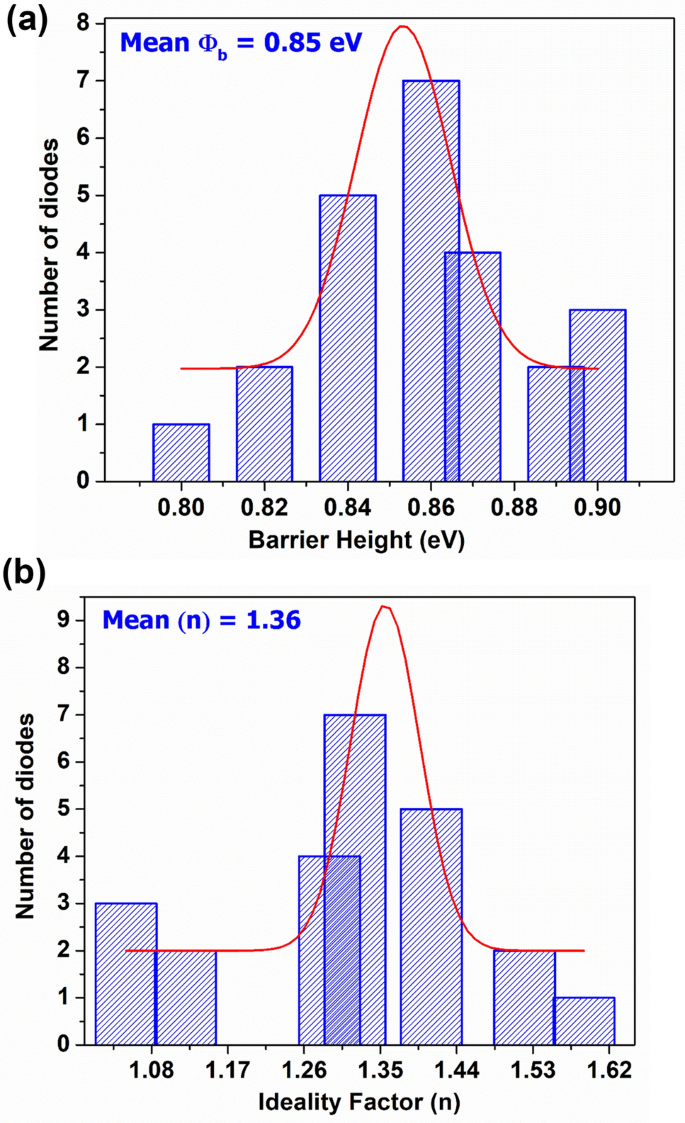
This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.ĭata Availability: All relevant data are within the manuscript and its Supporting information files.įunding: RK, FF and LFA were supported by NSF NeuroNex Award DBI-1707398 and The Gatsby Charitable Foundation. Received: FebruAccepted: JanuPublished: February 9, 2022Ĭopyright: © 2022 Khajeh et al. In addition to simulations, we provide a mean-field analysis to illustrate the properties of these networks.Ĭitation: Khajeh R, Fumarola F, Abbott L (2022) Sparse balance: Excitatory-inhibitory networks with small bias currents and broadly distributed synaptic weights. Interestingly, the speed, not the size, of synaptic fluctuations dictates the degree of sparsity in the model. Unlike standard balanced networks, these sparse balance networks exhibit robust nonlinear responses to uniform inputs and non-Gaussian input statistics. To support this activity, synaptic strengths must be drawn from high-variance distributions.
In these networks, irregular spontaneous activity is supported by a continually changing sparse set of neurons. The absence of experimental evidence for such large bias currents inspired us to examine an alternative regime that exhibits asynchronous activity without requiring unrealistically large feedforward input. The resulting excitatory-inhibitory (E-I) balance can generate spontaneous irregular activity but, in standard balanced E-I models, this requires that an extremely strong feedforward bias current be included along with the recurrent excitation and inhibition. Cortical circuits generate excitatory currents that must be cancelled by strong inhibition to assure stability.
